pH – ORP – Ledningsevne – Oksygen
Innen pH, ORP, ledningsevne og oksygen representerer vi flere anerkjente produsenter, som er spesialisert mot sine markeder. Produktsortimentet strekker seg fra helt enkle løsninger til det mest avanserte.
Fra vår hovedleverandør Knick leverer vi et spekter av forskjellige transmittere, elektroder og sensorhus som kan tilpasses fra de enkle til de mest krevende applikasjoner.
Vi kan tilby automatiserte systemløsninger inkludert automatisk rengjøring og kalbrering av målesensor.
pH
En pH-sensor brukes typisk til å måle mengden alkalinitet og surhet i vann og andre væsker. pH-sensorer er et viktig verktøy for å sikre kvaliteten og øke sikkerheten på et produkt og de prosessene som foregår i et avløps- eller produksjonsanlegg.
pH-skalaen er normalt basert på verdier fra 0-14, og en stoff med pH-verdi 7 regnes som nøytralt. Løsninger med en pH-verdi høyere enn pH 7, blir regnet som alkaliske (baser), mens løsninger med en pH-verdi mindre enn pH 7, er definert som sure (syrer). Tannkrem har for eksempel vanligvis en pH-verdi på pH 8-9. Magesyre ligger derimot på rundt pH 1-2.
Forskjellen mellom et alkalisk og et surt stoff har stor betydning for en del prosesser og bedrifter: kjøletårn, kjeler, produksjonsprosesser innen f.eks. næringsmidler, legemidler eller der det trengs ultrarent vann; kontroll av svømmebassenger og ulike typer miljøovervåking.
Uansett hvilken applikasjon du bruker den til, vil en pH-sensor gjøre det mulig å holde pH-verdien på et nivå som er ideelt for prosessen. Dette kan være alt fra sterke syrer til basiske stoffer. Det finnes mange ulike typer pH-sensorer til væskemåling, blant annet kombinasjons-pH-sensorer, laboratorie-pH-sensorer, prosess-pH-sensorer og differensial-pH-sensorer.
ORP
ORP-måling (Oxidation Reduction Potential) brukes også til vannkvalitetstesting og viser om et stoff kan oksidere eller redusere et annet. Positive ORP-målinger indikerer oksiderende stoffer og negative målinger reduserende stoffer. ORP-sensorer består av to elektroder (referanseelektrode og ORP-elektrode) og måler ORP-nivåer basert på elektronutvekslingen mellom ORP-elektroden og løsningen, mens referanseelektroden gir en stabil basisverdi for målingen.
ORP-sensorer er f.eks. ideelle for å bestemme forurensningsnivået i vann; høy ORP indikerer at vannet er rent, mens lav ORP indikerer forurensning. ORP-sensorer brukes på ulike områder som vannbehandling, svømmebassenger og fjørfeforedling. I svømmebassenger måler ORP-sensorer for eksempel kloreffektiviteten mot bakterier. Ulike ORP-verdier gjenspeiler ulike forurensningsnivåer, og høyere verdier betyr at vannet er sterilisert. ORP-sensorer gir mer nyansert informasjon enn pH-sensorer, noe som bidrar til effektive vannbehandlingsstrategier.
Ledningsevne
Konduktivitetssensorer måler en løsnings evne til å lede strøm basert på ionkonsentrasjonen. Sensorene inneholder elektroder laget av f.eks. platina eller gull, og fungerer ved å sende elektrisk strøm gjennom løsningen og måle den resulterende ledningsevnen. Ionkonsentrasjonen og ionenes mobilitet bestemmer ledningsevnen. Konduktivitetssensorer består vanligvis av to eller fire elektroder, inkludert en referanse- og en måleelektrode, og ofte også integrerte temperatursensorer for å sikre nøyaktigheten, da temperaturforskjeller vil påvirke måleresultatet. Ledningsevnen måles normalt i milli-siemens per cm (mS/cm) og gjenspeiler ionkonsentrasjoner. Konduktivitetssensorer brukes i mange bransjer, fra overvåking av vannkvalitet til vurdering av kjemiske løsninger og cellehelse.
Oksygen
Det finnes mange typer oksygensensorer som er basert på tre forskjellige funksjonsprinsipper:
- En endring i lysintensitet og amplitude – lys avgitt av et fluorescerende materiale når det utsettes for oksygen. Såkalt optisk oksygensensor.
- En endring i bølgelengden fra lyd, lys eller et magnetfelt når oksygen passerer gjennom det.
- Polarografisk og galvanisk prinsipp, hvor oksygenet penetrerer gjennom et membran og reduseres til hydroksid i en elektrolyttløsning med anode og katode.
Oksygensensorer brukes i mange prosesser og bransjer, deriblant biologiske renseanlegg, bioreaktorer innen farmasøytisk, oksygennivåkontroll i øl i bryggerier, helse og medisin etc.
Nyheter
-

Nytt agentur – Pyxis Lab Inc.
Vi har nå fått et nytt agentur, Pyxis Lab. Inc.,…
-
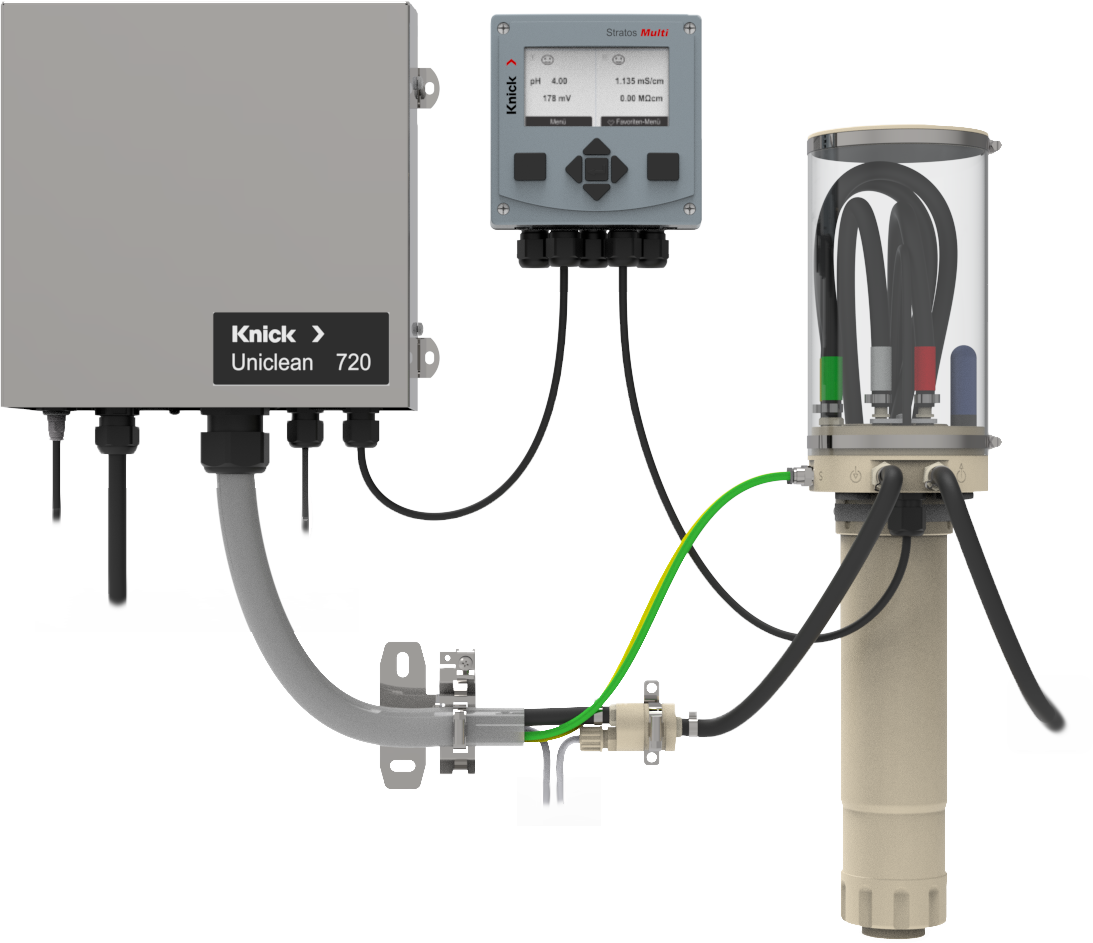
Nytt, fleksibelt sensorrengjøringssystem fra Knick
Uniclean 700-serien Moduloppbygget kontrollsystem for målesløyfer. Fleksibel elektropneumatisk kontroller for…
-

Raven-Eye radar flowmåler
J.S.Cock ute på serviceoppdrag for å løse kundens utfordringer.
-

OTD Bergen 2024
J.S.Cock med egen stand på Offshore Teknologidagene.